Environment & Energy
Showing Original Post only (View all)Multiscale Mechanistic Insights into Hydrogen Production from Microalgae via Molten Hydroxide-Mediated Thermochemical... [View all]
...Conversion.
The paper I'll discuss in this post is this one: Multiscale Mechanistic Insights into Hydrogen Production from Microalgae via Molten Hydroxide-Mediated Thermochemical Conversion Jun Li, Ling Lei, Dian Zhong, Hongyang Zuo, Han Zhu, Kuo Zeng, Haiping Yang, and Hanping Chen Environmental Science & Technology ]2026 60 (9), 7054-7066
It's written by scientists in China.
Since the paper involves the preparation of hydrogen from algae, one could argue, that it is literally "green" hydrogen, as chlorophyll is "green," although as we can easily see, the environmental sense of the word - which is widely abused, very, very, very often in connection with hydrogen scams - does not apply.
The um, introduction to the paper is rather cute:
Conventional thermochemical processes for microalgae, such as pyrolysis and gasification, are often constrained by persistent tar formation, ash clogging, and insufficient hydrogen yield/purity, (4−6) which adversely affect process efficiency and economic viability. In response, molten salt technology has emerged as a promising alternative, with molten hydroxides proving particularly effective. (7) These salts serve simultaneously as an efficient heat transfer fluid, an effective cracking catalyst, and a selective CO2 absorbent. (8,9) Previous work by the authors has demonstrated the superior effectiveness of molten hydroxides for microalgae processing, achieving a hydrogen yield of 67 mmol/g-microalgae with a purity of 80% while reducing tar formation by over 99%. (10,11) Approximately 58% of this hydrogen originated from the hydroxide itself, with the hydrogen utilization efficiency from the algae reaching 84.86%. This integrated performance outperforms that of the majority advanced catalytic and chemical looping systems in terms of clean and efficient hydrogen production...
During utilization, you don't say? Does this mean that we should ignore how hydrogen is made?
The IEA reports (as do many scientific papers) the carbon impact of hydrogen production:
IEA GHG Emissions of Hydrogen and Its Derivatives:
Perhaps they are discussing a perpetual motion machine, where the heat to melt group I hydroxides is provided by, um, hydrogen. Somehow I don't think so.
The molten hydroxide in this case is sodium hydroxide, mixed with sodium carbonate. The melting temperature of NaOH, a very caustic material, is 323 °C (598K). We may ask, whence the heat? (The authors reveal it at the end of the article by their affiliations.)
The experimental conditions:
Hastelloy is a nickel alloy with multiple other metals mixed in. It has been used in molten salt nuclear reactors, reactors I might note that have some rather serious materials science issues, particularly with respect to helium generation from neutron interactions with some of the lighter isotopes of nickel. (This is a problem.)
Some figures from the text:
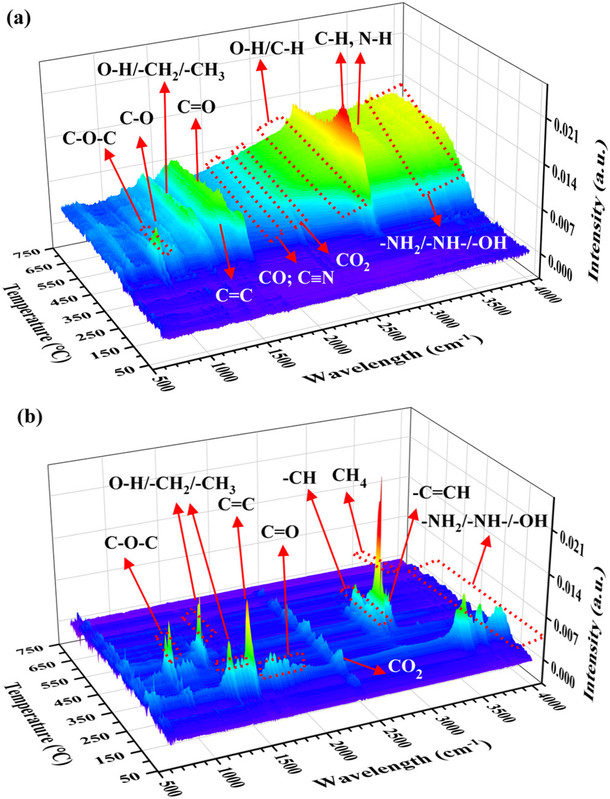
The caption:
This indicates that the hydrogen produced will not be pure and will contain multiple compounds, one of which is carbon monoxide. Mixtures of hydrogen and carbon monoxide are known as "syn gas" from which petroleum like mixtures can be obtained using "FT chemistry," Fischer-Tropsch (which was industrialized in Nazi Germany and in Apartheid era South Africa). However the nitrogenous compounds are likely to poison FT catalysts.
The lab scale reactor used in the experiments:
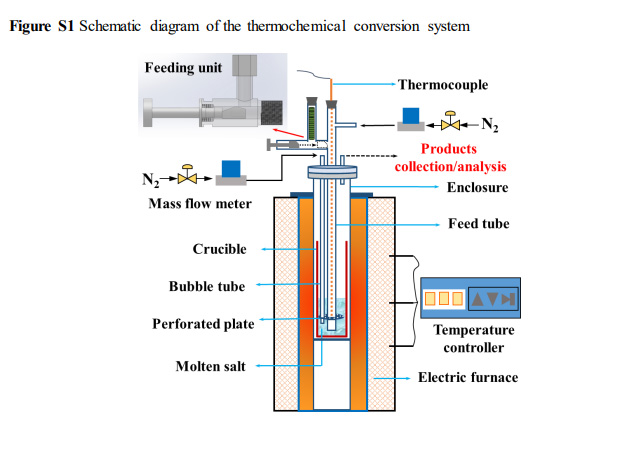
Note that the hydrogen is carried out of the molten hydroxides in a stream of nitrogen, meaning, again, that additional steps would be required to purify the already challenging hydrogen gas, a nightmare to store.
An additional note:
Benzene and toluene are both carcinogens, and account for the Proposition 65 notices on gasoline pumps in California:
On every gasoline pump in the State, the notice reads:
⚠ WARNING: Breathing the air in this area or skin contact with petroleum products can expose you to chemicals including benzene, motor vehicle exhaust and carbon monoxide, which are known to the State of California to cause cancer and birth defects or other reproductive harm. Do not stay in this area longer than necessary. For more information go to www.P65Warnings.ca.gov/service-station.
Of course, the fossil fuel marketeers trying to rebrand fossil fuels as "hydrogen" are only interested in cancer and carcinogenesis if they can stupidly tie it to nuclear plants.
Despite these warnings, people are willing to pay a lot of money for gasoline in California, more than ever, actually.
There is, by the way, a lot of nice chemistry in the paper, including a description of the PAHs - fossil fuel carcinogens - generated and the temperatures required to reduce, if not eliminate, them.
Usually, when citing scientific papers here, I often omit the institutions from which the authors come, but it seems important to do so in this case.
Ling Lei - State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, 1037 Luoyu Road, Wuhan, Hubei 430074, P. R. China
Dian Zhong - State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, 1037 Luoyu Road, Wuhan, Hubei 430074, P. R. China
Hongyang Zuo - State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, 1037 Luoyu Road, Wuhan, Hubei 430074, P. R. China
Han Zhu - State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, 1037 Luoyu Road, Wuhan, Hubei 430074, P. R. China
Haiping Yang - State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, 1037 Luoyu Road, Wuhan, Hubei 430074, P. R. China; Orcidhttps://orcid.org/0000-0002-8323-8879
Hanping Chen - State Key Laboratory of Coal Combustion, Huazhong University of Science and Technology, 1037 Luoyu Road, Wuhan, Hubei 430074, P. R. China
I added the bold.
This of course, is consistent with current Chinese practice for making hydrogen, using coal, something that is likely to be utilized more than ever for hydrogen production now that the Asian source of dangerous natural gas is constrained by the closing of the Straits of Hormuz has been closed by the stumbling insanity of the orange pedophile in the White House.
Thermochemical reactions for hydrogen production are well known, including the SI process that is now being piloted in China, driven by nuclear heat, which obviously is superior to coal except in the minds of German antinukes and, um, fossil fuel salespeople working to rebrand fossil fuels as "hydrogen" with slick commercial videos. The SI process does not require fossil fuels, and is catalytic water splitting via the generation of SO2 from the thermal decomposition of sulfuric acid followed by the iodine mediated Bunsen reaction.
Anyway, there's a lot of interesting stuff in this paper in a purely chemical mechanistic sense, and I've long had ideas about the use of molten hydroxides, although my interest is primarily in a highly radioactive hydroxide, cesium hydroxide, which has some very wonderful properties.
This said, this lab scale process, like much of the other hydrogen bullshit that flies around here, is not really useful.
Have a nice weekend.